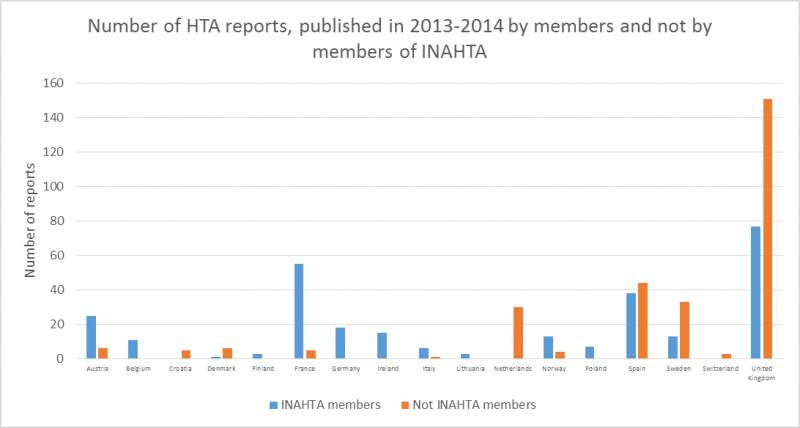
In total 301 out of 573 HTAs performed in 2013-2014 for medical devices in Europe were originated from non-INAHTA members. Countries with the most reports published by non-INAHTA members are Denmark (86%), Netherlands (100%), Spain (54%), Sweden (72%), Switzerland (100%) and United Kingdom (66%).
Health technology assessments are integral part of decision-making about utilization of medical devices in many European countries. Nevertheless, focus of industry and researchers is usually on large, often national-level HTA bodies (e.g. NICE in England, HAS in France, KCE in Belgium etc.).While assessments by national established bodies is important for decision-making at national level, this process may take years and usually limited number of technologies are getting into the focus for national evaluation. On the other side of the scale, there are hospital and regional health technology assessment units, which usually employ less rigorous methodology and perform assessment within few months. Obviously, results of local/regional evaluations have direct and immediate impact on reimbursement/purchase policy locally.
When it comes to monitoring of HTA activity, it is relatively easy to keep track of what is going on at the level of national HTA bodies. Although, it is difficult to scale up monitoring to the level of regional/hospital HTAs by the effort of a single company/institution. We offer a cost-effective solution to monitor activities, related to more than 100 national, regional and hospital HTA bodies in Europe. To illustrate significance of assessments by not usually considered HTA bodies, we have performed series of analysis using membership in international HTA associations, like International Network of Agencies for Health Technology Assessment (INAHTA) and EUnetHTA, as a proxy to the established role of HTA body in international community. In this post, we will focus on analysis for members/non-members of INAHTA.
INAHTA is a “network that connects HTA agencies to each other to support knowledge sharing and the exchange of information, and also to serve as a forum for the identification and promotion of other interests of HTA agencies”. To qualify for membership, organization shall have non-for-profit status and by funded by at least 50% by public sources.
In total 301 out of 573 HTAs performed in 2013-2014 for medical devices were originated from non-INAHTA members. Countries with the most reports published by non-INAHTA members are Denmark (86%), Netherlands (100%), Spain (54%), Sweden (72%), Switzerland (100%) and United Kingdom (66%).
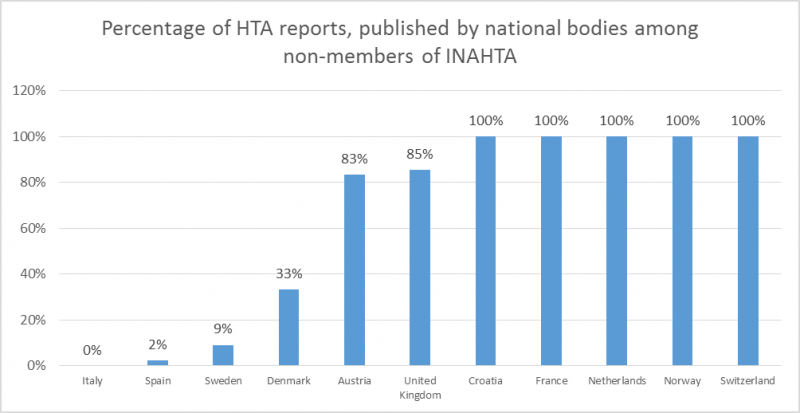
From non-INAHTA members from Croatia, France, Netherlands, Norway and Switzerland, only national HTA bodies produced reports related to medical devices. For other countries, the percentage of reports, produced by national bodies varied between 0% for Italy and 85% for the United Kingdom.
Focusing only on large national HTA bodies may limit ability to monitor HTA activities with direct, immediate impact on acceptance of technologies at local level.

